
AUGUST 6, 2025 AHMED A. METWALLY, STAFF RESEARCH SCIENTIST, AND A. ALI HEYDARI, RESEARCH SCIENTIST, GOOGLE RESEARCH play silent looping videopause silent looping videounmute videomute video
Leveraging wearable data and routine blood tests, we introduce a pioneering method for robustly predicting insulin resistance, establishing a scalable and accessible pathway for early type 2 diabetes risk assessment.
Type 2 diabetes impacts millions globally, with escalating prevalence. A primary precursor to this condition is insulin resistance (IR), where cellular response to insulin, vital for blood sugar regulation, is compromised. Early IR detection is paramount, as lifestyle modifications can frequently reverse it, preventing or delaying type 2 diabetes onset. However, current gold-standard IR measurement methodologies, such as the euglycemic insulin clamp or the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) requiring specific insulin assays, are often invasive, costly, or unavailable in routine check-ups. These factors erect substantial barriers to early detection and intervention, particularly for at-risk individuals unaware of their susceptibility.
Imagine harnessing readily available data, including wearable device information and common blood tests, to accurately estimate IR risk. In “Insulin Resistance Prediction From Wearables and Routine Blood Biomarkers,” we present a suite of advanced machine learning models capable of predicting IR using wearable data (e.g., resting heart rate, step count, sleep patterns) and routine blood tests (e.g., fasting glucose, lipid panel). This approach demonstrates exceptional performance across the studied population (N=1,165) and an independent validation cohort (N=72), especially in high-risk demographics, including individuals with obesity and sedentary lifestyles. Furthermore, we introduce the Insulin Resistance Literacy and Understanding Agent, a prototype powered by the state-of-the-art Gemini family of LLMs, designed to enhance comprehension of insulin resistance and facilitate interpretation and safe, personalized recommendations. This research paves the way for early identification of type 2 diabetes risk, enabling timely implementation of preventative strategies. Note: The models, predictions, and the Insulin Resistance Literacy and Understanding Agent are intended solely for informational and research purposes.

Metabolic subphenotypes of type 2 diabetes. Chronic insulin resistance, a precursor to approximately 70% of type 2 diabetes cases, stems from a combination of obesity, inactivity, and genetic predisposition.
Predicting IR using digital biomarkers & routine blood tests
We initiated the WEAR-ME study to explore the predictive power of readily available data for insulin resistance (specifically HOMA-IR). To streamline routine blood biomarker collection, we collaborated with Quest Diagnostics. 1,165 participants nationwide enrolled in the WEAR-ME study via the Google Health Studies app, a secure platform for digital research. The study received Institutional Review Board (IRB) approval, and all participants provided electronic informed consent and HIPAA Authorization. The cohort exhibited diversity in age, gender, geography, and BMI, with a median BMI of 28 kg/m², median age of 45 years, and a median HbA1c of 5.4%. Participants authorized the sharing of the following data: Wearable Data: Pseudonymized data from Fitbit or Google Pixel Watch devices (resting heart rate, step count, sleep patterns) to protect privacy. Routine Blood Biomarkers: Results from routine tests (fasting glucose, insulin, lipid panel) obtained during in-person visits to Quest Diagnostics for this research. Demographics and Surveys: Basic information and health questionnaires (age, weight, height, ethnicity, race, gender, self-reported health, diet, and medical history) completed at study initiation and conclusion. Utilizing this comprehensive multimodal dataset (the “WEAR-ME data”), we developed and trained deep neural network models to predict HOMA-IR scores, assessing the accuracy of estimating this key IR marker using various data combinations.

Illustration of our proposed modeling pipeline for predicting HOMA-IR and interpreting results with the Insulin Resistance Education and Understanding Agent.
Our findings, measured by the area under the receiver operating characteristic curve (auROC), reveal that integrating data streams substantially improved prediction accuracy over single-source utilization: Wearables + Demographics exhibited predictive capability (auROC = 0.70) for classifying IR. Incorporating Fasting Glucose with Wearables + Demographics significantly enhanced performance (auROC = 0.78), highlighting the impact of this routine blood test. Wearables + Demographics + Routine Blood Panels yielded optimal results, accurately predicting HOMA-IR values (R² = 0.50) and effectively classifying individuals with IR (auROC = 0.80, Sensitivity = 76%, Specificity = 84%, with HOMA-IR ≥ 2.9 indicating insulin resistance).

Left: Performance evaluation of IR prediction (classification). Right: Visualization of the precision-recall curve for selected feature sets. Average values are color-coded, with gray areas indicating standard deviation across five folds.
Crucially, our analysis shows that features derived from wearable data, such as resting heart rate, consistently emerged as highly significant predictors, alongside BMI and fasting glucose. This feature importance underscores the value of capturing lifestyle-related physiological signals.
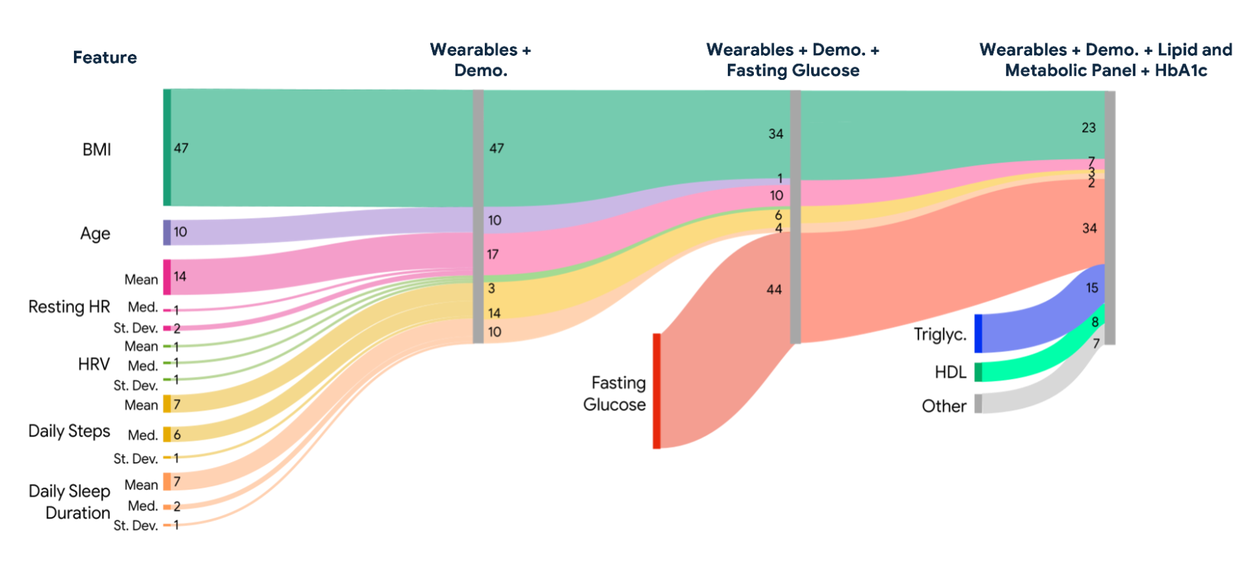
Sankey diagram illustrating relative feature importance (SHapley Additive exPlanations [SHAP] values) for each proposed nonlinear XGBoost model for direct regression.
Focusing on high-risk groups
Recognizing the heightened vulnerability of individuals with obesity and sedentary lifestyles to type 2 diabetes, we specifically assessed our model's performance within these subgroups: Obese Participants exhibited enhanced accuracy compared to the general population (sensitivity = 86% vs. 76%). Sedentary Participants showed higher accuracy than the obese subpopulation (sensitivity = 88%). Obese and Sedentary Participants demonstrated notably strong performance in this critical group (sensitivity = 93%, adjusted specificity = 95%; adjusted specificity here prioritizes minimizing misclassification of insulin-sensitive individuals). These experimental results suggest our approach effectively identifies individuals who could most benefit from early lifestyle interventions.

Results of classification performance across various lifestyle stratifications.
Validation and generalizability
To confirm our findings' robustness beyond the initial dataset, we applied our best-performing model (trained on WEAR-ME data) to an independent validation cohort (N=72). This cohort, recruited through a separate IRB-approved study, involved participants sharing wearable data via the Fitbit Charge 6 and providing blood biomarker data at a study center in San Francisco. This cohort presented a median BMI of 30.6 kg/m² and a median age of 44.5 years. Our validation results demonstrate that the trained models sustained strong predictive performance (sensitivity = 84%, specificity = 81%), underscoring their potential generalizability. However, as this remains a research prototype, its safety and effectiveness for any health-related application are yet to be established.

Overview of the independent validation cohort study, comparing model accuracies from the initial training/testing cohort with the external validation cohort to demonstrate potential generalizability.
Beyond prediction: Towards understanding and proactive steps
play silent looping videopause silent looping videounmute videomute videoIllustration of the proposed agentic architecture leveraging the HOMA-IR prediction model to assess insulin resistance risk for user education.
Predicting IR risk is crucial, but how can this information be rendered understandable and actionable? We explored integrating our prediction models with LLMs to empower individuals in comprehending their metabolic health. We developed the Insulin Resistance Literacy and Understanding Agent (IR Agent), built upon the advanced Gemini LLM family. Upon receiving a question about metabolic health, the IR Agent delivers personalized, contextualized educational answers grounded in the individual's study data and predicted IR status. With explicit user consent, the agent accesses specific, user-provided data points, retrieves current information, and performs calculations. Critically, interactions with the models or the IR Agent are intended for demonstration purposes, illustrating how such tools can assist users in exploring their results for informational and educational benefit.
Five board-certified endocrinologists evaluated responses from the IR Agent against a baseline model, strongly preferring the IR Agent's output for its comprehensiveness, trustworthiness, and personalization. This validates the potential of integrating predictive health models with LLMs to enhance public health understanding.

Overview of the Insulin Resistance Literacy and Understanding Agent (IR Agent). Left: Illustration of the proposed IR agent. Right: Evaluation results (win rate) of the IR agent against the base model as assessed by endocrinologists.
Conclusions and future work
Our research confirms that machine learning models integrating accessible wearable data and routine blood biomarkers can effectively predict insulin resistance, a critical precursor to type 2 diabetes. This approach offers significant advantages: Accessibility: Utilizes data readily available to most individuals. Early Detection: Identifies risk even before abnormal blood sugar levels manifest; notably, we identified numerous normoglycemic (HbA1c < 5.7) participants in our study already exhibiting IR. Scalability: Presents a potentially more scalable screening method than specialized IR tests. Personalization: Demonstrates robust performance in high-risk subgroups and holds potential for integration into personalized health platforms. This work unlocks possibilities for earlier, more accessible type 2 diabetes risk screening, facilitating timely lifestyle interventions to prevent or delay disease progression, especially for unaware individuals. Future endeavors include longitudinal model validation, assessing intervention impacts, incorporating genetic and microbiome data, and refining models for specific populations to ensure equitable performance across diverse groups. We firmly believe this research trajectory offers substantial promise for proactive and personalized metabolic health management.
Disclaimer
While our proposed approach, including the IR Agent, shows potential for diverse health applications, this research specifically addresses the critical need for early insulin resistance detection. The models discussed are not presented as approved medical devices or solutions. The models and the IR Agent are not medical devices and have not undergone clearance, approval, or review by the U.S. Food and Drug Administration (FDA) or any other regulatory body. This work is not a substitute for professional medical advice, diagnosis, or treatment. Real-world deployment necessitates rigorous testing, validation, and regulatory authorization.
Acknowledgements
The research presented is a collaborative effort between Google Research and partner teams, including: Ahmed A. Metwally, A. Ali Heydari, Daniel McDuff, Alexandru Solot, Zeinab Esmaeilpour, Anthony Z. Faranesh, Menglian Zhou, David B. Savage, Conor Heneghan, Shwetak Patel, Cathy Speed, and Javier L. Prieto. Google partnered with Quest Diagnostics, a leader in diagnostic information, enabling eligible participants to share their biomarker data from a free blood draw, which encompassed a comprehensive metabolic panel, cholesterol, triglyceride, and insulin level measurements.


